
Preparation of a minocycline polymer micelle
thermosensitive gel and its application in spinal
cord injury
Jun Gu,
ab
Xiaohu Cai,
ac
Faisal Raza,
d
Hajra Zafar,
d
Bo Chu,
b
Haitao Yuan,
b
Tianqi Wang,
b
Jiapeng Wang
e
and Xiaojun Feng*
ab
Neuroprotection is an important approach for the treatment of spinal cord injury (SCI). Minocycline (MC),
a known neuroprotective agent, has been utilized for SCI treatment, but its therapeutic e
ff
ect is limited
by instability and low bioavailability. Herein, we developed an innovative micellar thermosensitive
hydrogel (MCPP-M-gel) that encapsulates MC in polyethylene glycol (PEG)
–
poly(lactide-
co
-glycolic
acid) (PLGA) micelles to enhance its therapeutic e
ffi
cacy in a rat model of SCI. The micelles were
synthesized
via
the thin-
fi
lm hydration method and characterized for encapsulation e
ffi
ciency, particle
size, zeta potential, and polydispersity index (PDI). MCPP-M-gel demonstrated favorable physico-
mechanical properties and extended MC release over 72 hours
in vitro
without cytotoxic e
ff
ects on
neural crest-derived ectoderm mesenchymal stem cells (EMSCs). Thereafter, MC, MCPP-M, MCPP-M-gel
and a blank micellar thermosensitive gel were injected into the injured site of SCI rats. Histopathological
evaluation demonstrated that MCPP-M-gel could promote neuronal regeneration at the injured site of
the SC after 28 days. Immuno
fl
uorescence techniques revealed that MCPP-M-gel increased the
expression of neuronal class III
b
-tubulin (Tuj1), myelin basic protein (MBP), growth-associated protein
43 (GAP43), neuro
fi
lament protein-200 (NF-200) and nestin as well as reduced glial-
fi
brillary acidic
protein (GFAP) expression in damaged areas of the SC. In conclusion, this study innovatively developed
MCPP-M-gel based on a PEG
–
PLGA copolymer as a biomaterial, laying a solid foundation for further
research and application of MCPP-M-gel in SCI models or other neurodegenerative diseases.
Introduction
The spinal cord (SC) can be injured through mechanical
trauma, which induces a cataclysmal condition of neurons. This
is commonly known as spinal cord injury (SCI), which can have
an enormous socio-economic impact on a
ff
ected individuals. It
is a condition that can trigger a cascade of consequences such
as the generation of free radicals, perturbation of ionic balance,
vascular alterations, response to in
ammation and apoptosis.
1
The aforementioned events are degenerative and normally
associated with neuronal loss and axonal devolution, culmi-
nating in progressive damage to neuronal tissues and SC
functional impairment.
2
In recent years, scientists have
explored preclinical models of SCI to investigate numerous
drugs that have the potential to repair injured tissue and
enhance the recovery of SC functions.
3,4
From a preclinical point
of view, the complex nature of SCI has hampered the successful
development of therapeutic interventions for this condition.
This is because only few of them have been translated into
clinical settings, albeit with limited success, probably because
only one aspect of SCI has been targeted.
5
Hence, for e
ff
ective
treatment of SCI, prospective drugs should be developed to
target multiple aspects of the injury to facilitate neuro-
protection, neuronal regeneration and recovery of SC function.
Besides, long-term use of conventional medications for SCI
treatment can cause various drug-related medical problems for
patients. Notable among these medical problems are dyslipi-
demia, hypertension, anxiety/depression, osteoarthritis and
osteopenia/osteoporosis.
6
Novel therapeutic strategies are
required for the e
ff
ective treatment of SCI. At present, neuro-
protective medications that block sodium channels (riluzole)
and act as antioxidants (vitamin E) and anti-in
ammatory
agents (curcumin, methylprednisolone and minocycline),
among others, have been widely explored to treat SCI.
7
As a derivative of tetracycline, minocycline (MC) is considered
an antibiotic that is available clinically with non-antibiotic
bene
cial e
ff
ects such as anti-apoptosis, neuroprotection, anti-
oxidation and anti-in
ammation.
8
Because of its neuro-
protective potential, MC has widely been used to treat disorders
a
School of Medicine, Yangzhou University, Yangzhou, Jiangsu 225009, China. E-mail:
featherinwind81@163.com
b
Department of Orthopedics, Xishan People's Hospital, Wuxi, Jiangsu 204105, China
c
Department of Rehabilitation, Xishan People's Hospital, Wuxi, Jiangsu 204105, China
d
School of Pharmacy, Shanghai Jiao Tong University, Shanghai 200240, China
e
School of Pharmacy, Jiangsu University, Zhenjiang, Jiangsu 212013, China
Cite this:
DOI: 10.1039/d4na00625a
Received 29th July 2024
Accepted 15th September 2024
DOI: 10.1039/d4na00625a
rsc.li/nanoscale-advances
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Nanoscale
Advances
PAPER
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

of neurons: Alzheimer's disease, amyotrophic lateral, multiple
sclerosis, Parkinson's disease and SCI.
9
–
11
In particular, MC
demonstrated
the
potential
to
increase
levels
of
anti-
in
ammatory cytokines and reduce pro-in
ammatory factors in
animal models of SCI.
12
In another work, the authors showed that
MC decreased the apoptotic rate in oligodendrocytes and
neurons by stimulating the production of microglial pro-nerve
factor, inhibiting activated caspases and suppressing mitochon-
drial cytochrome c release.
13,14
Evidently, scientists have discov-
ered that the cascade of secondary apoptosis that occurs a
er
acute SCI plays vital roles in SCI pathogenesis.
15
In this same
work, the authors showed that Bcl-2 plays a very important role in
the death of nerve cells a
er SCI.
15
In particular, Bcl-2 is
considered an anti-apoptotic protein that can inhibit or e
ff
ec-
tively prevent the apoptosis caused by various pathways a
er SCI
by promoting the repair of injured nerve tissue and neuro-
protection.
16
Indeed, Bcl-2 plays a neuroprotective role in SCI by
inhibiting neuronal cell death. Currently, studies have shown
that MC intervention could inhibit neuronal apoptosis by
increasing Bcl-2 expression, thereby playing a therapeutic role in
SCI.
17
More importantly, the highly lipophilic nature of MC may
facilitate the drug to readily cross the blood
–
brain barrier and
subsequently distribute into the tissues of the central nervous
system and cerebrospinal
uid.
18
Nonetheless, clinical applica-
tions of MC are limited by its systemic toxicity
19
and possibly its
inability to penetrate the blood
–
spinal cord barrier, which
prevents e
ff
ective delivery of conventional drugs to the SC,
especially when administered systemically, orally or into the
peridural space.
20,21
To overcome the above challenges, scientists
through preclinical studies have explored the potential of nano-
materials, such as micelles and hydrogels, to e
ff
ectively target
drugs to the injured site of SC. The nanotechnological-based
delivery system of drugs has various advantages, such as
increased aqueous solubility, decreased toxicity, enhanced bio-
logical availability and prolonged circulation time in the blood-
stream.
22
Most lipophilic drugs have been encapsulated using
poly(lactate-
co
-glycolic acid) (PLGA), a polymer that has been
approved by the Food and Drug Administration (FDA) because it
o
ff
ers advantages, such as increased bioavailability of drugs,
reduced toxicity of the system and good biodegradability.
23
Nonetheless, PLGA applications are limited because of the easy
opsonization and rapid clearance of polymer-based drugs by the
reticulo-endothelial system (RES) a
er intravenous administra-
tion. Therefore, formulation scientists usually conjugate hydro-
phobic PLGA with hydrophilic polyethylene glycol (PEG) to
fabricate a co-polymeric (PLGA
–
PEG) block platform that is
amphiphilic. Importantly, PEGylation of PLGA-based nano-
particles resulted in minimal neurotoxicity and enhanced
stability of bioactive deoxyribonucleic acid (DNA) in primary
neurons of the hippocampus.
24
Besides, PLGA
–
PEG co-polymer
has been employed by other scientists to fabricate thermosensi-
tive nanoplatforms for applications in SCI.
25
In an aqueous milieu,
amphipathic polymers can assemble to spontaneously produce
nanostructured polymeric micelles, which have the advantages of
increased physical stability, prolonged release, targeted delivery,
greater bioactivity, solubilization of active compounds or high
stability of the incorporated drug.
26
The available literature has
posited that polysialic acid-based polymeric micelles loaded with
MC could substantially lower the formation of scars and reduce
injury to nerve and myelin sheaths through anti-in
ammation
and anti-oxidation.
27
In another study, polymeric micelles devel-
oped with scar tissue targeting ability peptides, such as cysteine
–
alanine
–
glutamine
–
lysine (CAQK), could treat SCI through the
speci
c delivery of apocynin to lesion tissue.
28
Biodegradable
amphipathic micelles were developed by Wang and colleagues,
29
which enhanced the synergistic neuroprotective activity of dexa-
methasone acetate and glucocorticoid in the SCI model.
Notwithstanding the potential of these formulations, their short-
falls hamper their clinical applications in SCI. Speci
cally, the
instability of polymeric micelles in the blood causes the rapid
release of MC a
er administration, which a
ff
ects the accumula-
tion of the drug in the injured site of SC.
30,31
Thus, the search for
ideal nanocarriers of MC to target the drug to SC is ongoing.
As a category of polymers, hydrogels absorb water to form
a 3-dimensional network through their hydrophilic groups,
which have the morphology of
exible tissue with similarity to
the extracellular matrix features.
32,33
Common biomaterials,
such as hydrogels, signi
cantly a
ff
ect SCI treatment because
they are biocompatible with some of them displaying electrical
conductivity that is compatible with tissues of SC.
34
Hydrogels
have been employed to incorporate several cells or active
compounds and drugs with the potential to treat SCI because of
their greater capability to deliver drugs in a prolonged fashion
to target sites.
34
Besides, the plasticity and high biocompati-
bility nature of hydrogels can facilitate the accumulation of the
polymers at the defective SC and the continuous release of
drugs, such as MC.
34
More importantly, scientists have reported
the potential of hydrogels to support the growth of axons and
their regeneration.
35
Thus, scientists can leverage the advan-
tages of micelles and hydrogels to design nanocarriers that are
more compatible with biological stimuli.
Recently, scientists have renewed their interest in the
in situ
formation of hydrogel, which occurs as a result of the trans-
formation of aqueous polymeric solutions into gels due to
alterations in milieu conditions, such as pH and temperature.
36
It has been posited that this type of hydrogel can provide several
advantages compared with conventional ones when they are
formed under conditions of physiology and maintain their
integrity for a desired period. In recent times, researchers have
developed a thermosensitive micellar-hydrogel (MH) system
that displays the advantages of both polymeric micelles and
hydrogels, which has attracted much interest in tissue engi-
neering.
37
Thus, this system was developed to achieve sustained
and long-lasting delivery of hydrophobic drugs
via
the forma-
tion of a hybrid micelle-cross-linked hydrogel, which potentially
serves as a reservoir of lipophilic solutes.
38
Particularly, the self-
assembling
of
hydrophilic
–
hydrophobic
co-polymer
into
hydrogel occurs in an aqueous environment and under physi-
ological conditions, wherein it can
ow like micelles in
a concentration-dependent fashion.
39
Various studies have
indicated that the MH system has been developed with di
ff
erent
materials for various applications. For instance, Qin and
colleagues developed an MH system for the sustained delivery
of celastrol and its application in renal
brosis.
38
In another
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

work, Sun and co-authors fabricated an MH system that
demonstrated the increased anti-breast cancer e
ff
ect of quer-
cetin.
40
In addition, Xu and co-experimenters fabricated a ther-
mosensitive MH system to incorporate and deliver docetaxel to
the intra-tumoral site.
41
Additionally, Yi
et al.
developed an MC-
loaded hydrogel with F127 as the polymer for application in
periodontal diseases.
42
As one of the most broadly utilized
thermos-reversible gelling polymers for hydrogel development,
the PEG-PGLA block has been found to demonstrate excellent
biocompatibility and biodegradability in terms of delivery of
drugs.
43
Based on previous studies, this study sought to improve the
therapeutic e
ff
ect of MC
via
the development of MC-loaded
PEG
–
PLGA micelles (MCPP-Ms) before they were developed
into an
in situ
thermosensitive MC-loaded gel (MCPP-M-gel) for
the treatment of SCI. Thus, this work sought to provide new
ideas for the development and utilization of MC.
Materials and methods
Materials
Sinopharm Chemical Reagent Co., Ltd., supplied acetonitrile,
ethanol, methanol, potassium chloride, ammonium acetate,
P188 and P407. The phosphate bu
ff
er saline (PBS), sodium
chloride, para-formaldehyde, 2% phosphotungstic acid and
hematoxylin
–
eosin (HE) staining kit were provided by Beijing
Solarbio Technology Co., Ltd. The minocycline (99% purity),
glucan gel G-50, coumarin 6 and ethylenediaminetetraacetic
acid disodium salt (EDTA-2Na) were purchased from Sigma-
Aldrich (St. Louis, MO, USA). PEG3000
–
PLGA3000 was
provided by Toyongbio (Shanghai, China).
Analytical method for determining MC content
To detect the MC content, a high-performance liquid chro-
matographic (HPLC) technique was employed, which was
mainly composed of a Waters Symmetry C18 (5
m
m, 4.6 mm
×
250 mm) column with the eluent being bu
ff
er solution of
ammonium acetate (obtained
via
dissolution of ammonium
acetate-15 g, potassium chloride-10 g and ethylenediamine
tetraacetate-5 g prior to dilution to 1000 mL) and acetonitrile at
a ratio of 70 : 30. Other chromatographic conditions for the
analysis were detection wavelength (280 nm), column temper-
ature (25 °C),
ow rate (1.0 mL min
−
1
) and injection volume (20
m
L). The establishment of an
in vitro
MC standard curve showed
a good linear relationship at a concentration range of 5
–
100
m
g
mL
−
1
with the following linear equation:
y
=
22910
x
−
131687
(1)
(
R
2
=
0.9970, where
y
denotes peak area and
x
represents MC
concentration). The retention time for MC was 5.5 min (Fig. 1A).
Preparation of MCPP-M
Based on previous studies,
44
the thin
lm hydration method was
used to prepare blank micelles and MCPP-M (PEG3000
–
PLGA3000 50 : 50). Through preliminary studies, the drug
loading proportion was determined based on investigations of
particle size and polydispersed index (PDI) of the micelles with
di
ff
erent drug contents. Based on preliminary studies (data not
shown), di
ff
erent ratios of MC : PEG
–
PLGA were investigated:
1 : 8, 1 : 9, 1 : 10, 1 : 11 and 1 : 12 (Table 1). Besides, a blank
micelle was prepared with a particle size of 43 nm and a poly-
dispersed index (PDI) of 0.217.
To prepare MCPP-M, MC and PEG
–
PLGA were accurately
weighed into a round glass bottle before 30 mL of chromato-
graphic ethanol was added to dissolve the mixture completely.
A
erward, the bottle containing the solution was rotated to
evaporate at 45 °C until a uniform thin
lm was formed. Under
reduced pressure, rotavap was employed for 12 h to remove the
residual organic solvent. Later, 10 mL of puri
ed water was
added to hydrate the
lm and allowed to stand for 20 min
before it was
ltered through 0.45
m
m membrane
lters to
remove impurities and obtain MCPP-M. Notably, the best
preparation was selected using the particle size, PDI and
encapsulation rate.
Characterization of MCPP-M
NanoBrook 90 Plus particle size analyzer equipped with
dynamic light-scattering (DLS) and phase-analysis light-
scattering (PALS) techniques were employed to accordingly
measure the zeta potential, particle size and PDI of MCPP-M
based on existing method and conditions.
45
Speci
cally, the
MCPP-M was appropriately diluted before measurement (in
triplicate) of the above-mentioned physical characteristics at
a 90° angle and temperature of 25 °C.
Morphological observation of MCPP-M using the
transmission electron microscopic (TEM) technique
Observation of the morphology of MCPP-M was carried out a
er
dilution of the sample to approximately 200
m
g mL
−
1
with
deionized water. Later, one drop of dried sample was placed on
a copper net prior to staining with 2% phosphotungstic acid,
and subsequent detection with TEM.
Estimation of encapsulation rate and drug loading of MCPP-
M
Based on a modi
ed existing method,
46
the encapsulation rate
and drug loading of MCPP-M were detected a
er the separation
of the micellar system with glucan gel chromatography. Brie
y,
the glucan gel G-50 (50 g) was placed into the chromatography
column before the water balance. A
erward, the micelle (1 mg
mL
−
1
) was carefully added to the column and eluted with
puri
ed water. Later, the sample (10 mL each) was collected
before the content of the encapsulated MC was detected with
HPLC. When MC could no longer be detected in the eluted
puri
ed water portion, a 0.2% sodium chloride solution was
used for the detection of MC, which was considered the unen-
capsulated MC. In addition, the drug loading was measured
through the following procedure: a certain concentration (1 mg
mL
−
1
) of MCPP-M was placed in a tube and centrifuged for
30 min at 3700 rpm before the supernatant was removed and
analyzed with HPLC. The calculation of the encapsulation rate
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a
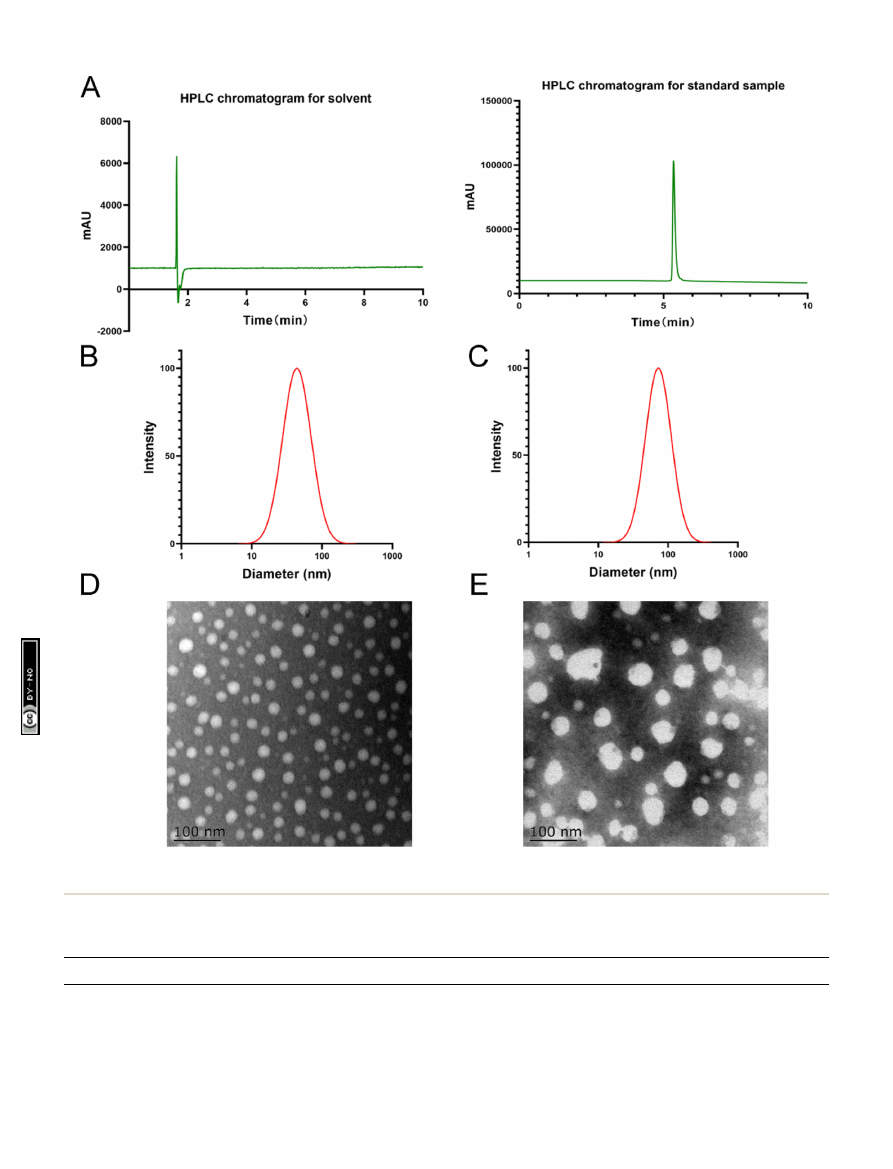
Fig. 1
Chromatographic analysis of minocycline (MC) and physical characterization of MC-loaded micelles (MCPP-M). (A) HPLC chromatogram
for MC. Particle size distribution of (B) blank micelles and (C) MCPP-M. TEM micrographs of (D) blank micelles and (E) MCPP-M.
Table 1
Characteristics of di
ff
erent preparations of minocycline (MC) micelles (
n
=
3, mean
±
standard deviation)
Ratios
Particle size (nm)
Polydispersed index (PDI)
Encapsulation rate (%)
1 : 8
162.32
1.520
0.302
0.006
63.21
2.780
1 : 9
129.11
2.050
0.283
0.008
72.39
2.190
1 : 10
72.16
1.060
0.227
0.005
89.38
1.840
1 : 11
61.83
1.080
0.221
0.006
90.18
2.010
1 : 12
59.81
1.290
0.219
0.004
91.65
1.150
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

and drug loading was based on the following existing eqn (2)
and (3):
46
Encapsulation rate
ð
%
Þ ¼
Q
encapsulated
Q
total
100%
;
(2)
Drug loading
ð
%
Þ ¼
Q
encapsulated
Q
lipids
100%
;
(3)
where
Q
encapsulated
denotes the quantity of MC encapsulated in
the micelle,
Q
lipids
denotes the total quantity of lipid content in
the micelle and
Q
total
represents the total quantity of drug in the
dispersed micelle.
Testing the storage stability of MCPP-M
Based on a modi
ed method in a previous study,
47
the prepared
MCPP-Ms were kept in a closed container at 25 °C and 4 °C for
one month, wherein the micelle was sampled on days 0, 15 and
30 within the period to observe the physical appearance, while
the particle size distribution, PDI and zeta potential were
determined to test the stability of the formulation.
Testing the
in vitro
release pro
le of MCPP-M
Testing the
in vitro
release behavior of unencapsulated MC and
MCPP-M was performed using dialysis method
48
with three
bu
ff
er solutions [PBS (pH 7.4 and 6.8) and hydrochloric acid
(HCl, pH 1.2)] as release media. A
erward, an aliquot (1 mg) of
MC (solubilized in a very small amount of methanol) and
MCPP-M (dissolved in 1 mL of puri
ed water) were placed in
a dialysis bag (MV
=
3500 dalton). A
er the two ends of the
dialysis bags had been tied, the bags were placed in an Erlen-
meyer
ask containing 100 mL of the release media prior to
oscillation (at 37 °C, 100 rpm). A
erward, samples (1 mL each)
were collected at pre-determined intervals (10, 30, 45, 60, 90,
120, 180, 240, 360, 480, 600, 720, 1440, 2160, 2880, 3600 and
4320 min), while 1 mL of fresh release media were added
simultaneously. Later, the samples were diluted with methanol
in accordance with the HPLC method described above. The
cumulative MC release was carried out while considering the
replenishment of the media,
49
and the entire measurements
were performed in triplicate prior to the calculation of the
average MC concentration.
Uptake of MCPP-M by neural-crest-derived ectoderm
mesenchymal stem cells (EMSCs)
A previously developed
uorescence microscopic technique
50
was utilized to ascertain the uptake of MCPP-M by the EMSC
cells. In brief, the EMSCs were seeded at 1
×
10
5
cells per well
density on coverslips in Dulbecco's Modi
ed-Eagle's Medium
(DMEM, Wisent Biotech., Co., Ltd., Nanjing, China). A
er they
had grown 70% con
uence, the medium was replaced with
a fresh medium composed of FITC labeled BCPP-M at a
nal
MC concentration of 10
m
g mL
−
1
before incubation at various
times (1, 2, 4 and 8 h). Later, PBS was used to wash the cells
twice before
xing them with para-formaldehyde (PFA, 4%) and
subsequent staining of the nucleus for 5 min with 4
0
,6-di-
amidino-2-phenyl-indole (DAPI, 0.5
m
g mL
−
1
) in the dark at
room temperature. The cells were observed with a
uorescence
microscope (Labophot-2, Nikon, Melville-NY, USA) a
er the
coverslips had been mounted on a glass slide.
Preparation and characterization of MCPP-M-gel
The MCPP-M-gel was prepared using the method described in
a previous study
41
with slight modi
cations. Concisely, 5 mL of
MC
–
PEG
–
PLGA was accurately weighed in water (10 mg mL
−
1
).
It was then poured into test tubes pre-supplemented with 0.18 g
of P188, 0.54 g of P407 and 1 mL of 0.003 g per mL chitosan
glacial acetic acid solution. A
er complete mixing, the mixture
was naturally expanded at 4 °C and stored overnight to remove
air bubbles, a
er which a transparent solution was formed to
obtain the MCPP-M-gel. Notably, the above procedure was fol-
lowed to prepare a blank micellar thermosensitive gel that did
not contain MC.
Morphological observation of MCPP-M-gel using the scanning
electron microscopic (SEM) technique
As described elsewhere,
41
a dehydrated MCPP-M-gel was
lyophilized before observation of the morphology with SEM (S-
3400, Hitachi, Japan) a
er the gel was coated with gold. Both
MCPP-M-gel and blank thermosensitive gel were visualized
using the appropriate technique.
Determination of gel formation temperature (GFT) and
gelation time (GT) of MCPP-M-gel
The inverted test tube method
51
was employed to determine the
GFT of the MCPP-M-gel. In brief, an appropriate volume of
MCPP-M-gel was placed in the transparent tube. The sample was
placed under a constant temperature water bath and subjected to
slow heating starting from 30 °C at a temperature amplitude of
1 °C, coupled with temperature insulation of 10 min. Later, the
tube was inverted, and the
ow of the gel was observed to
determine whether the gel was formed. A
erward, the gel was
removed and placed in a 4 °C refrigerator. A
er the sol was
Table 2
Stability of MCPP-M after storage for 30 days at 4 °C and 25 °C (
n
=
3, mean
±
standard deviation)
Period
PDI
Zeta potential (mV)
Particle size (nm)
4 °C
25 °C
4 °C
25 °C
4 °C
25 °C
0 day
0.227
0.005
0.227
0.005
−
28.31
0.900
−
28.31
0.900
72.16
1.060
72.18
1.140
15 days
0.229
0.006
0.229
0.008
−
28.16
0.930
−
27.09
0.970
73.12
1.250
74.29
1.610
30 days
0.229
0.007
0.232
0.009
−
27.46
0.980
−
26.62
1.120
73.68
1.850
76.47
1.950
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a
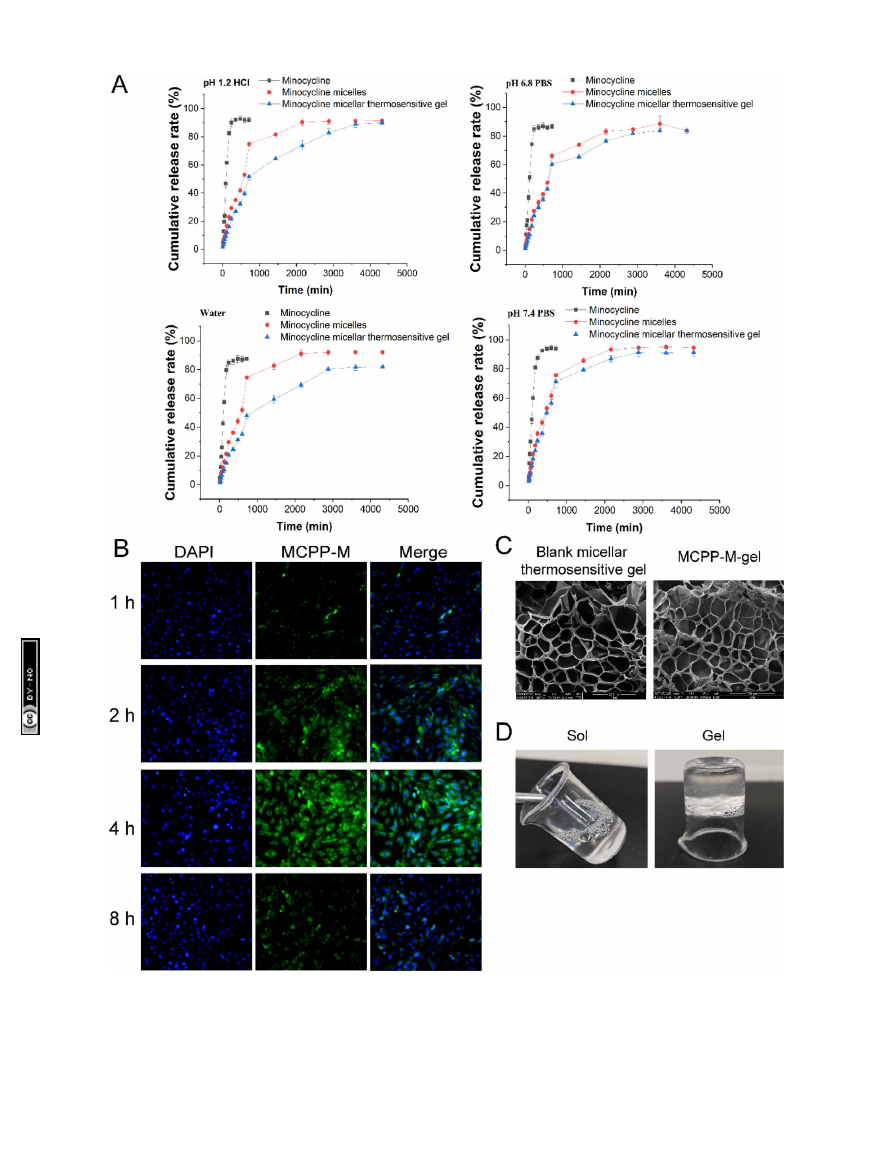
Fig. 2
Physical characterization of MCPP-M and MC-micellar thermosensitive hydrogel (MCPP-M-gel). (A)
In vitro
release of MC from free MC,
MCPP-M and MCPP-M-gel. (B) E
ff
ect of MCPP-M uptake by neural-crest-derived ectoderm mesenchymal stem cells (EMSCs) (20
×
). (C) SEM
micrograph of blank micellar thermosensitive gel and MCPP-M-gel. (D) Digital photos of MCPP-M-gel changed from sol to gel.
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a
restored, the gel was placed in a water bath. Later, the heating
interval was narrowed, and the gel temperature (
T
g
) was accu-
rately determined a
er estimating the approximate gel temper-
ature. This procedure was repeated three times, and the average
was calculated.
Regarding the measurement of GT, a procedure described in
previous works
51,52
was employed. Brie
y, the transparent tube
lled with 2 mL of MCPP-M-gel was placed in a 37 °C constant
temperature water bath to observe a change in the mixture.
When the colorimetric tube was inverted and the solution was
no longer
owing, the solution was considered to form
a hydrogel.
Determination of erosion rate and rheological pro
le of
MCPP-M-gel
The
in vitro
erosion rate of MCPP-M-gel was determined at 37 °C
using a membrane-free system, as stated elsewhere.
53
The
empty bottle (10 mL cillin bottle) was weighed, and MCPP-M-gel
(about 2 g) was added before placement in a mechanical shaker
(37 °C, 150 cycles per min) for 10 min so that the polymer
solution could completely form the gel. A
erwards, the gel was
removed, and 1 mL of PBS (pH 7.4) was added as the release
medium before the sample was placed into a water bath with
a constant temperature oscillator, which oscillated at 37 °C and
a rate of 30 rpm. Later, the release medium was completely
discarded at the set time, and the bottle was dried prior to
weighing. Then, the water bath was equilibrated for 10 min and
supplemented with 1 mL of PBS solution (37 °C). This proce-
dure was repeated until the remaining gel was less than 10% of
the starting amount. Three parallel groups were set for each
experiment. Then, erosion times were recorded, and the average
was calculated.
To determine the rheological pro
le of the MCPP-M-gel,
a strain-controlled rheometer was utilized for the experiment.
The apparatus utilized the parallel plate method, wherein the
diameter was roughly 25 mm, whereas a space of 1.00 mm was
maintained between the parallel and Peltier plates. Later,
a frequency sweep test was employed to determine the elastic
modulus (
G
0
) and viscous modulus (
G
00
). At a rate of 1 °C min
−
1
,
the plate was heated from 10 °C to 45 °C while maintaining the
frequency at 10 rad s
−
1
. A
erward, the sol or gel behavior was
evaluated as
G
0
<
G
00
(sol) or
G
00
<
G
0
(gel) of the hydrogel system.
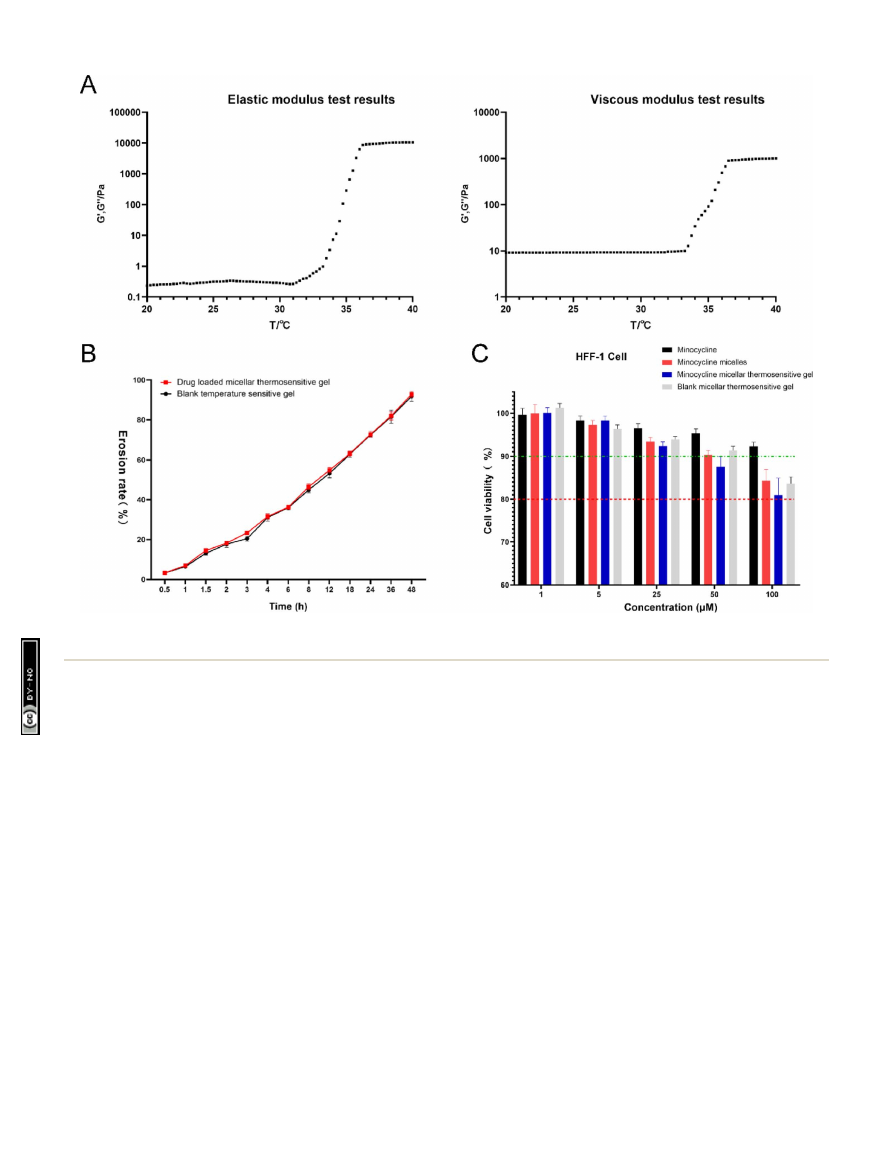
Fig. 3
Minocycline (MC)-loaded micellar thermosensitive hydrogel (MCPP-M-gel) characterization. (A) Elastic and viscous moduli of MCPP-M-
gel. (B) Erosion rate of MCPP-M-gel. (C) Biosafety of MCPP-M-gel.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a
Testing of MCPP-M-gel e
ff
ect on cell viability
Prior to their use in cytotoxicity studies, the Chinese Academy of
Sciences Stem Cell Bank benevolently supplied human foreskin
broblast cells (HFF-1), which were cultured based on ideas
from previous studies.
54
Concisely, culturing of the cells under
appropriate conditions (37 °C and 5% CO
2
) was performed with
DMEM (Wisent Biotech., Co., Ltd., Nanjing, China) comprising
penicillin
–
streptomycin (PS, 1%, Gibco, Thermo-Fisher Scien-
ti
c, Waltham, MA, USA) and fetal-bovine serum (FBS, 15% heat
inactivated, Bioagrio, Mountain View, CA, USA). Only cultured
cells that had reached the lag growth stage were used for the
experiment. The cells were seeded into 96 well plates (100
m
L per
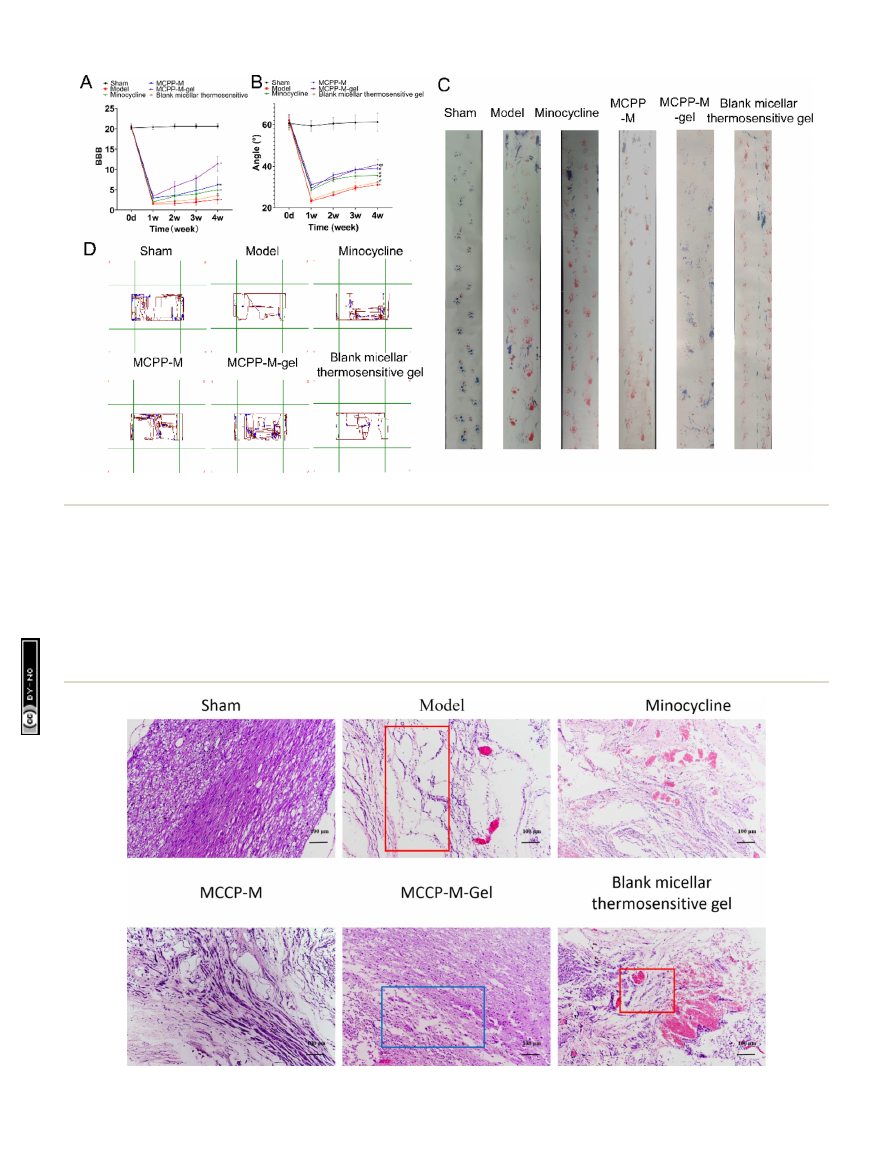
Fig. 4
Functional behavior assessment. (A) BBB score. (B) Slanted-plate experiment. (C) Foot-print inspection. (D) Open
fi
eld experiment.
Fig. 5
Histopathological observation (10
×
).
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a
well) under conditions such as density (3
×
10
5
cells per well)
and medium per well (2
m
L) before incubation under the same
above-mentioned conditions. The following four groups were
studied: MC, MCPP-M, MCPP-M-gel and blank micellar ther-
mosensitive
gel.
Using
3-[4,5-dimethyl-thiazol-2-yl]-2,5-
diphenyl-tetrazolium bromide (MTT) assay, the cells were co-
cultured with varied concentrations (1, 5, 25, 50 and 100
m
M)
of the above-mentioned dosage forms. Subsequently, incuba-
tion was performed for 1
–
4 h under the same aforementioned
conditions a
er the addition of MTT solution (10
m
L) to each
well of the above-mentioned plate. For each experiment, at least
ve replicates were set up for each group. Before measuring the
absorbance at 570 nm with a microplate reader, the crystals of
formazan were dissolved in a solubilization solution (dimethyl
sulfoxide). Later, cell viability was calculated based on an
existing equation.
55
Testing of the
in vitro
release behavior of MCPP-M-gel
In testing for the
in vitro
release behavior of MCPP-M- gel, the
same method, releasing media, conditions, apparatus and
procedures were employed as described above for MCPP-M.
Surgery of rats for construction of SCI model and
administration of MCPP-M-gel
The Sprague-Dawley (SD) rats (with an average weight of about
250 g) were supplied by the Center for Laboratory Animal
Research at Jiangsu University (Zhenjiang, China). The rats
fasted the night before the surgery, and they were not given
Fig. 6
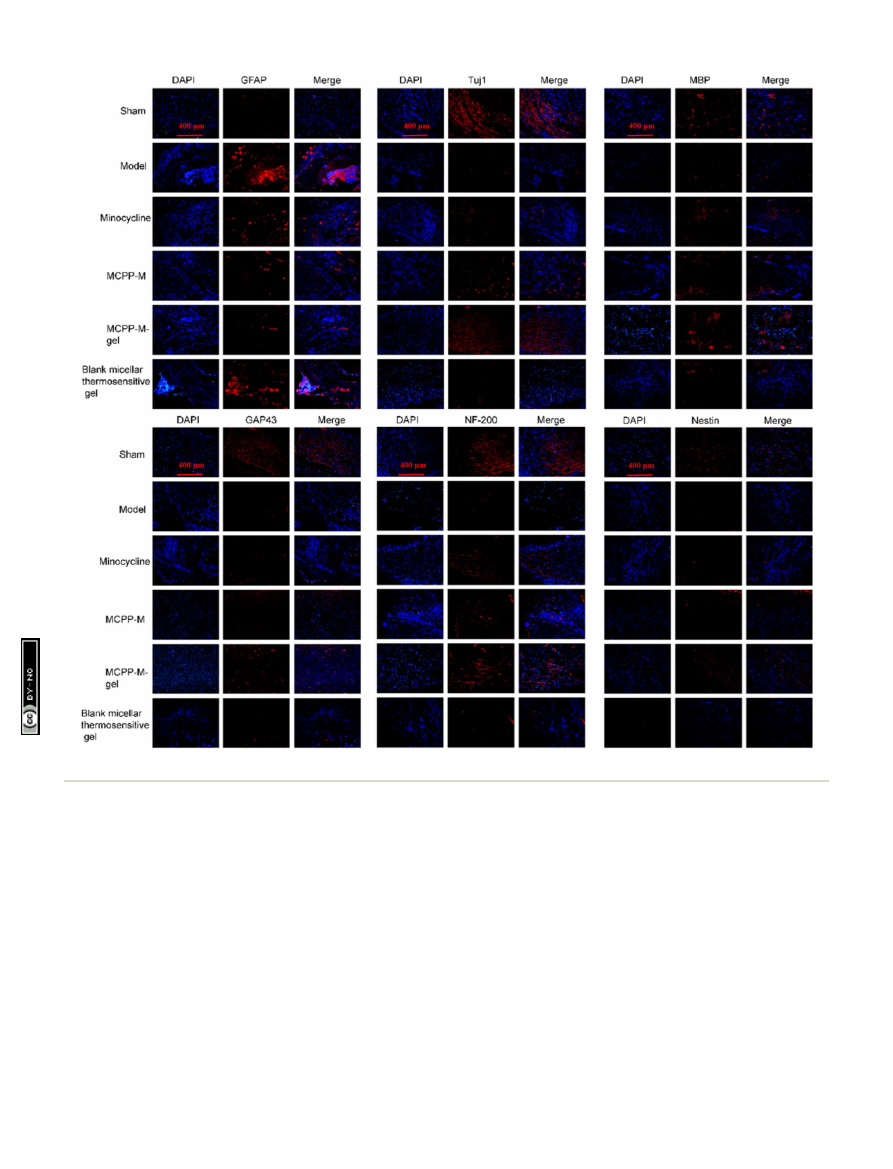
Expressions of GFAP, Tuj1, MBP, GAP43, NF-200, and nestin proteins were detected using immuno
fl
uorescence assessment (10
×
).
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

water on the surgery day. A
er ensuring that the animals were
well cared for in a conducive laboratory environment, approval
was obtained for the animal experimental protocol from the
Jiangsu University Ethics Committee for the Care and Use of
Laboratory Animals (UJS-IACUC-2023040801). The operative
surgery was performed as described elsewhere
56
with slight
modi
cations. In short, 7% chloral hydrate was employed for
the anesthesia of the rats a
er disinfection of the back skin of
the animal
via
the intraperitoneal route. Subsequently, the rats
were placed on an ultra-clean table for the operation process,
particularly by positioning the T12 vertebrae (located in the rat
of the highest point) a
er the midline back incision. Later, the
rats were opened layer by layer, and the muscles and vertebrae
were separated before forceps were used to remove the spine
and dura to expose the bone marrow. Ultimately, blade tran-
section was performed at the T10
–
11 segment spinal cord to
cause complete spinal cord transection.
The following groups (
n
=
10 rats in each group) were
analyzed: group A (sham rats), group B (model rats), group C
(rats that received MC), group D (rats that received MCPP-M),
group E (rats that received MCPP-M-gel) and group F (rats
that received blank micellar thermosensitive gel). Treatment
was carried out as follows. Rats in groups C, D, E and F received
their respective dosage forms at a dose of 90 mg kg
−
1
(ref. 57)
once daily for 1 week
via
intra-peritoneal injection, followed by
the application of MCPP-M-gel and blank micellar thermo-
sensitive gel to the injury site of SC; subsequently, a layer by
layer closure of the muscles and skin was performed. The rats
were subjected to good post-operative care, as established in
other works.
58
Post-injury motor behavior assessment
via
open
eld
evaluation
Open
eld motor function at 0, 3, 7, 14, 21 and 28 days was
assessed using the Basso, Beattie and Bresnahan (BBB) scoring
methods.
59
Each hindlimb was sorted by two blind observers
and simultaneously videotaped. The BBB score ranged from
0 (unchanged hind limbs) to 21 (normal gait) to assess func-
tional improvement a
er injury and treatment. Later, BBB was
performed in uninjured animals to determine whether these
materials had any early impairment of normal motor function.
Through the test device, the slope test was performed at 0, 3, 7,
14, 21 and 28 days. The maximum angle of the rats holding the
posture for 5 s without falling was recorded and averaged to
obtain a single score for each animal.
Behavioral investigation
All the rats were subjected to a series of behavioral analyses a
er
sciatic nerve injury. Restoration of locomotor activity in the le
hind limb was considered evidence of adequate regeneration of
muscle and recovery of function a
er sciatic nerve injury, which
was monitored by analyzing the pattern of free walking using
the method referenced in previous studies.
60
In this test, the rats
were placed on a track (500 mm long, 100 mm wide, covered
with white paper at the bottom and placed in a black box).
Meanwhile, they were allowed to walk with dark dye on the soles
of the hind paw before the analysis of their gait and recovery.
Histological evaluation
A
er 28 days, the histological observation was performed a
er
the preparation of appropriate solutions, namely sodium
pentobarbital (4 mg/100 g body weight), sodium chloride
(0.9%), and paraformaldehyde (PFA, 4%, 0.0 1 M) that was
bu
ff
ered with PBS (pH
=
7.4). Later, SC was collected from T8
–
T10 before the sample was
xed in PFA (4%) and embedded in
para
ffi
n. A
erward, hematoxylin
–
eosin (HE) staining was
carried out, and the lesion center was observed under a light
microscope.
Detection of relevant protein expression using
immuno
uorescence techniques
A previously established method
61
was followed to perform
immuno
uorescence techniques.
In performing the immuno
uorescent technique, the SC
sections were treated overnight with primary antibodies,
namely Tuj1 (1 : 100, Abcam, Cambridge, England), GAP43 (1 :
100, Abcam, Cambridge, England), GFAP (1 : 100, Abcam,
Cambridge, England), NF-200 (1 : 100, Cell Signaling Tech-
nology, USA), MBP (1 : 100, Abcam, Cambridge, England) and
nestin (1 : 100, Abcam, Cambridge, England), at 4 °C before
incubation at 37 °C for 2 h with HRP-conjugated secondary
antibodies. Prior to the observation of the SC sections using the
uorescent microscopic technique (Eclipse 80i, Nikon, Japan),
the sections were stained using an Immunol Fluorescence
Staining Kit (Beyotime, China).
Statistical analysis
Statistical analysis and construction of the experimental data
into graphs were performed with Graph-Pad Prism v8.0 so
ware
(USA). Comparison between two groups was carried out using
an unpaired
t
test, while more than two groups were compared
using a one-way analysis of variance (ANOVA). In terms of
statistical signi
cance level, at least
p
< 0.05 was considered
a
er the data were expressed as mean
±
standard deviation.
Results and discussion
Preparation and physical characterization of MCPP-M-gel
The MCPP-M-gel was successfully prepared using the thin
lm
hydration method, as described in the methods. During the
preparation of MCPP-M, di
ff
erent ratios of MC : PEG-PGLA co-
polymer (Table 1) were explored. From Table 1, the physical
characteristics of the above-mentioned ratios (1 : 8 to 1 : 12)
ranged from 59.81
±
1.29 to 162.32
±
1.52 nm (particle size),
0.219
±
0.04 to 0.302
±
0.01 (PDI) and 63.21
±
2.78 to 91.65
±
1.15% (encapsulation rate). It was observed that the particle size
and PDI of the micelle decreased, while the encapsulation rate
increased with an increase in the concentration of the PEG-PGLA
co-polymer. Based on the stability of MCPP-M, a formulation with
an MC : PEG-PGLA ratio of 1 : 10 was selected for subsequent
studies. The preparation had a relatively smaller size coupled
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

with a high zeta potential and encapsulation rate. In comparison
with blank micelles (particle size of 43
±
0.11 nm and a PDI of
0.217), the MCPP-M had a micellar size of 72.16
±
1.060 nm, PDI
of 0.227
±
0.005, zeta potential of
−
28.31
±
0.900 mV, encap-
sulation rate of 89.38
±
1.840% and drug loading of 8.08
±
0.08%. The observed smaller hydrodynamic diameter of blank
micelles than MCPP-M may be owing to the incorporation of
a large drug molecule such as MC (457.48 g mol
−
1
) within the
micellar core.
62
However, the structure of the MCPP-M-gel will be
comprehensively investigated in the future to potentially under-
stand the actual structure of the hydrogel. Besides, the negative
sign of the zeta potential of MCPP-M may be ascribed to free
carboxylic ends of PLGA moiety in the co-polymer.
63
Further, MCPP-M displayed increased loading of MC, which
may be attributed to its high lipophilic nature. Usually, hydro-
phobic drugs, such as MC, favorably interact with the lipophilic
moiety of PEG-PGLA polymers, thereby increasing the loading
e
ffi
ciency of such drugs. Additionally, the encapsulation rate of
MCPP-M may be increased by the bulky nature of the PEG-PGLA
polymer
64
via
reduction of net sheer stress induced by increased
particle size a
er MC incorporation.
65
In terms of micellar
morphology, MCPP-M particles were observed to be spherically
distributed with any sign of agglomeration, which relatively
agreed with that of blank micelles. Collectively, MCPP-M
exhibited reasonably increased zeta potential and encapsula-
tion rate, which are important attributes for the subsequent
development of the hydrogel.
Storage stability and
in vitro
release pro
le of MCPP-M
Because micelles tend to disintegrate and release entrapped
drugs prematurely, this phenomenon can decrease the e
ffi
-
ciency of drug delivery and potentially create concerns about
toxicity.
66
Therefore, it is essential that the excipients of the
preparation contribute immensely to the stability of the
micelles. As described in the methodology, the stability of
MCPP-M was investigated for 30 days at 4 °C and 25 °C. The
results are depicted in Table 2. Physical characteristics such as
PDI, zeta potential and particle size of MCPP-M were evaluated,
which were found to be stable throughout the studied period.
Although these characteristics altered slightly, they did not
reach a statistically signi
cant level. Thus, the developed MCPP-
M demonstrated appropriate stability, which is essential for
in
vivo
application. It is possible that the stability of MCPP-M may
be due to the PEG-PGLA polymer,
67
which has previously been
used to stabilize lipophilic molecules, such as MC.
To simulate the
in vivo
environment, we investigated the
in
vitro
release pattern of MC from MCPP-M using the di
ff
usion
technique. Fig. 2 shows the result of the
in vitro
cumulative
release pro
le of MCPP-M. Compared to free MC (Fig. 2A), the
cumulative MC release from MCPP-M at 72 h demonstrated
a prolonged release pattern (Fig. 2A). In particular, at 12 h, free
MC released the drug at signi
cantly increased cumulative rates
in all the four media compared to MCPP-M [HCl pH 1.2 (95.52
±
2.00%
vs.
77.94
±
2.00%,
p
=
0.0011), PBS pH 6.8 (86.64
±
1.55%
vs.
66.21
±
1.64%,
p
=
0.0001), water (87.47
±
1.98%
vs.
74.46
±
2.81%,
p
=
0.0028) and PBS pH 7.4 (94.01
±
1.71%
vs.
75.65
±
3.51%)]. The observed fast release rate of MC within
12 h corroborated previous studies.
68
This phenomenon may be
attributed to the increased solubility of MC in an aqueous
medium.
69
Beyond 12 h, the amount of MC released from free
MC could not be detected in the four media. However, MCPP-M
released MC in a sustained-release manner until 72 h, wherein
maximum cumulative release rates in the four media were
achieved in an order of PBS pH 7.4 (94.55
±
1.48%) > water
(92.07
±
1.25%) > HCl pH 1.2 (91.25
±
0.90%) > PBS pH 6.8
(83.93
±
2.14%). Overall, the
in vitro
release of MC was pro-
longed by the micellar system for 72 h, which may have
improved the delivery of the drug to the injured area of the SC.
Uptake of MCPP-M by EMSCs
A
uorescence microscopic technique was employed to ascer-
tain the potential absorption of MCPP-M
in vitro
, wherein the
micelles were labeled with green-
uorescent FITC dye before
incubation with EMSCs. From the results displayed in Fig. 2B, at
1 h, the MCPP-M could be observed to accumulate in the nuclei
(as indicated
via
blue DAPI dye) of EMSCs. However, MCPP-M
labeled with FITC was taken up by macrophages and the cyto-
plasm of EMSCs began a
er 2 h of incubation. Later, nearly all
MCPP-M could be taken up by EMSCs a
er 4 h of incubation
but subsequently declined a
er 8 h. This phenomenon may be
attributed to the transport of MCPP-M into other endocytic
intermediates in EMSCs.
70
Collectively, these
ndings suggest
the rapid internalization of MCPP-M by EMSCs
via
the e
ff
ective
intrinsic action of the cell. The e
ff
ect of the physical charac-
teristics of MCPP-M on cellular uptake and the mechanism
underlying the uptake of the micelles by EMSCs was not studied
in this work. Because these parameters are important for
scientists to understand the targeted delivery behavior of MCPP-
M, our subsequent research work will prioritize this limitation.
Preparation, gelation characteristics and erosion rate of
thermosensitive MCPP-M-gel
Based on the challenges of micelles, a thermosensitive micellar
hydrogel of MC was prepared and characterized. A
er the
successful development of the thermosensitive MCPP-M-gel, its
transformation from sol to gel was observed using the inverted
test tube method. Through SEM micrographs of the cross-
section of the lyophilized MCPP-M-gel, an irregular micropo-
rous structure of the gel (Fig. 2C) was observed. In terms of
gelation formation, it was discovered that a
er the MCPP-M-gel
was heated (to 37 °C), it changed from sol to gel (Fig. 2D). In
contrast, MCPP-M-gel changed from gel to sol a
er the
temperature decreased to room temperature (25 °C). Addition-
ally, the GFT and GT of MCPP-M-gel were determined to be
35.13
±
0.31 °C and 101.77
±
3.95 s, respectively. Because the
average GFT of MCPP-M-gel was closer to the physiological
temperature (37 °C), this suggests that the hydrogel could
potentially be transformed from sol to gel under physiological
conditions a
er injection. A rheological test was employed to
further a
ffi
rm the transformation of MCPP-M-gel from sol to
gel. As illustrated in Fig. 3A, increased elastic modulus (
G
0
,
G
00
)
and viscous modulus (
G
0
,
G
00
) were observed a
er the
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

temperature had risen to approximately 35 °C. Both elastic and
viscous modulus were similar, indicating that the phase-
transition temperature of the MCPP-M-gel was around 35 °C.
Overall, these results imply that MCPP-M-gel demonstrated
thermally reversible behavior. Moreover, the erosion rate for
48 h was determined. The results are displayed in Fig. 3B. The
erosion rate usually entails depletion of the polymeric material,
which, in this case, was PEG-PGLA co-polymers. It was observed
that blank micellar thermosensitive gel and MCPP-M-gel eroded
gradually and increased over time, achieving a maximum rate
(92.84%) at 48 h (Fig. 3B). Overall, these
ndings suggest the
successful preparation of MCPP-M-gel, which demonstrates
acceptable physico-mechanical properties.
Biosafety and
in vitro
release of MCPP-M-gel
Cell death degree and cell proliferation suppression a
er
chemical exposure are measured through cytotoxicity assays.
Because novel nanoparticles are to be applied clinically, it is
relevant to determine their cytotoxic e
ff
ects on the biological
environment. In this regard, the cytotoxic e
ff
ects of four dosage
forms (namely MC, MCPP-M, MCPP-M-gel and blank micellar
thermosensitive gel) were ascertained
in vitro
using HFF-1 cells.
Fig. 3C depicts the e
ff
ects of MC, MCPP-M, MCPP-M-gel and
blank micellar thermosensitive gel on HFF-1 cell viability. It was
observed that the cell viability of HFF-1 decreased as the
concentration of the dosage forms increased. At higher
concentrations (50
m
M) of the dosage forms, the cell viability of
HFF-1 was still above 80%, suggesting that the above-
mentioned dosage forms were biocompatible, and hence can
potentially be applied in the clinical settings.
71
The
in vitro
study
of the dosage forms provides only a rapid generation of
knowledge on their biosafety. This investigation lacks an
in vivo
toxicological outcome, which is very crucial for the validation of
the formulations. Despite this limitation,
in vitro
biosafety
results can be employed as predictors prior to the validation
phase of toxicological evaluation. In the not-too-distant future,
our group will comprehensively investigate the mechanism
underlying the interaction of the above-mentioned dosage
forms with biological systems.
The
in vitro
release pro
le of MC from MCPP-M-gel was also
evaluated using the same method described for the micelle. In
general, the MCPP-M-gel exhibited a prolonged release of MC.
The release of the drug started as early as 10 min (approximately
3%) in all the media and continued until 72 h. The rates of
cumulative release of MC from MCPP-M-gel in the four media at
72 h were in the following order: PBS pH 7.4 (91.25
±
2.90%) >
HCl pH 1.2 (89.86
±
2.38%) > PBS pH 6.8 (83.81
±
1.87%) >
water (81.94
±
2.07%). However, the di
ff
erence between the
total amount of MC released from MCPP-M (94.55
±
1.48%) and
MCPP-M-gel (91.25
±
2.90%) was statistically insigni
cant, with
the amount of the drug released by the former being slightly
higher than that of the latter. This seeming di
ff
erence may be
owing to the potential of hydrogels to control the release of
drugs in a prolonged fashion,
72
which has great prospects for
possible clinical applications in the di
ffi
cult-to-reach areas
within the human body.
E
ff
ect of MCPP-M-gel on SCI
Treatment for complex SCI entails the regeneration of neurons
and functional recovery. The prevention of neuronal cell death
is usually a major aim of neuroprotection, which is achieved by
restricting the processes of secondary injury that occur minutes,
hours and days a
er the incidence of primary injury. Few
successes have been achieved in using traditional treatment
strategies for secondary injury in SCI. Hence, a novel approach
to incorporating small molecules into biomaterials that have
the potential to promote the long-lasting delivery of drugs and
neuroprotection has the prospect of treating SCI. The MH
system exhibits the above-mentioned characteristics very well.
Hence, the MH system was utilized to incorporate a known
neuroprotective agent, such as MC (MCPP-M-gel). A
erward,
the potential of MCPP-M-gel to promote neuronal regeneration
was evaluated in a rat's SCI model.
E
ff
ect of MCPP-M-gel on functional recovery from SCI
The BBB score and slanted-plate experiment were employed to
measure functional recovery. Then, the e
ff
ect of MC, MCPP-M,
MCPP-M-gel and blank micellar thermosensitive gel on the
functional recovery of SCI rats for four weeks was observed.
Fig. 4A and B illustrate the therapeutic e
ff
ect of MC, MCPP-M,
MCPP-M-gel and blank micellar thermosensitive gel
via
BBB
locomotion score and slanted-plated experiment, respectively.
In terms of BBB scoring, there was a gradual increase in the BBB
scores of the rats in each group throughout the 28 day post-SCI
period. Notably, a
er 28 days, the rats that received MCPP-M-gel
substantially attained a behavioral score of 11.4 compared to
other groups: model (2.6), MC (5.0), MCPP-M (6.2) and blank
micellar thermosensitive gel (3.6). The experimental inspection
of rat foot-print presents rats' gradual movement ability
between distinct groups and di
ff
erences in trajectories of hind
limbs (Fig. 4C) throughout the study periods a
er treatment. In
this regard, it was observed that the rats in the sham group
dragged and de
ected their hind limbs. Through foot-print
inspection, nearly consistent trajectories of the hind-limbs of
rats were observed in the MCPP-M-gel groups, amid improve-
ment in the extent of tripping and swaying, although the rats in
each group struggled to support their weight for movement of
their hind-limbs. The treatment e
ff
ect of MCPP-M-gel may be
ascribed to the long-lasting release of MC,
73
which may have
promoted adequate regeneration of muscle and recovery of
function a
er sciatic nerve injury.
E
ff
ect of MCPP-M-gel on neuronal regeneration
Histologically, the e
ff
ect of MCPP-M-gel on neuronal protection
through histopathological evaluation was observed using HE
staining. The results of HE staining (Fig. 5) demonstrated
divergences in terms of morphology within distinct groups. In
particular, neuronal di
ff
erences were observed. In the untreated
groups, the SCI generally depicted tissues that were larger,
hydrolysable, loosely structured, and coupled with neuronal
and nuclear. Three days a
er SCI, it was observed that rats in
the untreated group exhibited substantial shrinking of
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

morphology with the presence of disordered structures and
increased reduction in neuronal numbers (as shown in the red
boxes in Fig. 5 model and blank micellar thermosensitive gel).
Importantly, the SCI of rats that received MCPP-M-gel displayed
a signi
cant reduction in the damaged area a
er 28 days of
treatment, amid re-organization of the injured sites and
neuronal retain (as shown in the blue boxes in Fig. 5 MCPP-M-
gel). These results collectively suggest that MCPP-M-gel could
promote neuronal regeneration at the injured site of the SC,
which corroborates existing works.
74
It is possible that MCPP-M-
gel was quickly transformed to hydrogels
in situ
under the
physiological conditions at the injured site of the SC, which
delivered MC in a sustained manner for improvement in the
neuroprotective activity of MC.
Further con
rmation of the neuroprotective potential of
MCPP-M-gel was carried out by performing an immuno
uo-
rescence technique using markers, such as GFAP, Tuj1, MBP,
GAP43, NF-200 and nestin (Fig. 6). As a marker of astroglial
injury, GFAP is used to ascertain the incidence and severity of
traumatic SCI. Additionally, Tuj1 is a neuronal marker for the
identi
cation of neural di
ff
erentiation.
75
Similarly, MBP is
considered a marker of injury to brain tissue; hence, it is used in
the diagnosis of neurological diseases.
76
As a phosphoprotein of
axons, the GAP43 is regarded as an indicator for the growth of
axons and synapses, which is increasingly expressed during
traumatic SCI.
77
Additionally, NF-200 can be used as an index of
large alpha
–
beta
ber neurons that are myelinated.
78
Finally,
nestin is a protein marker of neuroprogenitor, especially that of
the hippocampus, which can be developed into astrocytes and
neurons.
79
One way or the other, these proteins play an
important role in neuronal regeneration. Hence, a potential
neuroprotective agent, such as MCPP-M-gel, should potentially
increase or lower its expression in SC tissues. Using the
immuno
uorescence technique, it was observed that MCPP-M-
gel treatment and other dosage forms the up-regulated expres-
sion of Tuj1, MAP43, NF-200, MBP, GAP43 and nestin but the
down-regulated expression of GFAP in SC tissues, while the
hydrogel showed better e
ff
ect.
This study did not conduct a more comprehensive evaluation
of the biocompatibility and immunogenicity of MCPP-M-gel, as
these characteristics may a
ff
ect the tolerance and e
ffi
cacy of the
implant. Although cell toxicity tests were conducted in this
experiment, the
in vitro
results may not be completely consistent
with the
in vivo
situation, and further in-depth studies are
required on the safety of MCPP-M-gel material. The materials
used in this experiment were prepared on a laboratory scale, and
the preparation process may be di
ffi
cult to scale up to industrial
production. Therefore, further optimization may be required in
the production process to facilitate the clinical application of this
technology. These are all areas that must be continuously
improved in subsequent experiments.
Conclusions
Herein, a micellar thermosensitive MC-loaded hydrogel with
PEG-PGLA co-polymer as the biomaterial (MCPP-M-gel) was
successfully developed for the treatment of SCI. The MCPP-M-
gel had acceptable physical characteristics, such as smaller
particles, narrow PDI and increased encapsulation rate, and it
sustainably released MC within 72 h. Evaluation of MCPP-M-gel
through
in vivo
studies revealed that the hydrogel promoted
neuroprotection and functional recovery in SCI rats by
increasing the expression of Tuj1, MAP43, NF-200, MBP and
nestin albeit reducing GFAP expression in damaged areas of
SCI. Thus, the incorporation of MC into MCPP-M-gel may have
potentially improved its neuroprotective e
ff
ect in SCI rats.
Data availability
Data will be available upon request to the corresponding
author.
Con
fl
icts of interest
No competing interest declared by authors.
Acknowledgements
This project was supported by 2022 General Program of Jiangsu
Provincial Health Commission (M2022026), 2022 Key Discipline
Innovation Team of Wuxi Health Commission (CXTD2021022),
General Program of Wuxi Health Commission (M202357) and
Wuxi Health Commission project (Q202215).
References
1 Z. Nazemi, M. S. Nourbakhsh, S. Kiani, Y. Heydari,
M. K. Ashtiani, H. Daemi and H. Baharvand,
J. Controlled
Release
, 2020,
321
, 145
–
158.
2 N. A. Silva, N. Sousa, R. L. Reis and A. J. Salgado,
Prog.
Neurobiol.
, 2014,
114
, 25
–
57.
3 J. Ruschel, F. Hellal, K. C. Flynn, S. Dupraz, D. A. Elliott,
A. Tedeschi, M. Bates, C. Sliwinski, G. Brook, K. Dobrindt,
M. Peitz, O. Brüstle, M. D. Norenberg, A. Blesch,
N. Weidner, M. B. Bunge, J. L. Bixby and F. Bradke,
Science
,
2015,
348
, 347
–
352.
4 N. Nagoshi, H. Nakashima and M. Fehlings,
Molecules
, 2015,
20
, 7775
–
7789.
5 J. Wilcox, D. Cadotte and M. Fehlings,
Neurosci. Lett.
, 2012,
519
, 93
–
102.
6 T. Patel, J. Millignan and J. Lee,
J. Spinal Cord Med.
, 2017,
40
,
54
–
61.
7 A. Ulndreaj, A. Badner and M. G. Fehlings,
F1000Research
,
2017,
6
, 1907.
8 R. Shultz and Y. Zhong,
Neural Regener. Res.
, 2017,
12
, 702.
9 Y. Choi, H. Kim, K. Shin, E. Kim, M. Kim, H. Kim, C. Park,
Y. Jeong, J. Yoo, J. Lee, K. Chang, S. Kim and Y. Suh,
Neuropsychopharmacology
, 2007,
32
, 2393
–
2404.
10 S. Zhu, I. G. Stavrovskaya, M. Drozda, B. Y. S. Kim, V. Ona,
M. Li, S. Sarang, A. S. Liu, D. M. Hartley, D. C. Wu,
S. Gullans, R. J. Ferrante, S. Przedborski, B. S. Kristal and
R. M. Friedlander,
Nature
, 2002,
417
, 74
–
78.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

11 K. Maier, D. Merkler, J. Gerber, N. Taheri, A. V. Kuhnert,
S. K. Williams, C. Neusch, M. B¨
ahr and R. Diem,
Neurobiol.
Dis.
, 2007,
25
, 514
–
525.
12 B. W. Festo
ff
, S. Ameenuddin, P. M. Arnold, A. Wong,
K. S. Santacruz and B. A. Citron,
J. Neurochem.
, 2006,
97
,
1314
–
1326.
13 Y. D. Teng, H. Choi, R. C. Onario, S. Zhu, F. C. Desilets,
S. Lan, E. J. Woodard, E. Y. Snyder, M. E. Eichler and
R. M. Friedlander,
Proc. Natl. Acad. Sci. U. S. A.
, 2004,
101
,
3071
–
3076.
14 T. Yune, J. Lee, G. Jung, S. Kim, M. Jiang, Y. Kim, Y. Oh,
G. Markelonis and T. Oh,
J. Neurosci.
, 2007,
27
, 7751
–
7761.
15 F. Abbaszadeh, S. Fakhri and H. Khan,
Pharmacol. Res.
, 2020,
160
, 105069.
16 S. Qian, Z. Wei, W. Yang, J. Huang, Y. Yang and J. Wang,
Front. Oncol.
, 2022,
12
, 985363.
17 J. He, J. Mao, L. Hou, S. Jin, X. Wang, Z. Ding, Z. Jin, H. Guo
and R. Dai,
Exp. Anim.
, 2021,
70
, 21
–
0028.
18 H.-S. Kim and Y.-H. Suh,
Behav. Brain Res.
, 2009,
196
, 168
–
179.
19 A. M. Martins, J. M. Marto, J. L. Johnson and E. M. Graber,
Antibiotics
, 2021,
10
, 757.
20 F. Pinelli, F. Pizzetti, V. Veneruso, E. Petillo, M. Raghunath,
G. Perale, P. Veglianese and F. Rossi,
Biomedicines
, 2022,
10
,
1673.
21 F. Rossi, G. Perale, S. Papa, G. Forloni and P. Veglianese,
Expert Opin. Drug Delivery
, 2013,
10
, 385
–
396.
22 Y. Hu, X. Chen, Z. Li, S. Zheng and Y. Cheng,
J. Biomed.
Nanotechnol.
, 2020,
16
, 54
–
64.
23 I. Khan, A. Gothwal, A. K. Sharma, P. Kesharwani, L. Gupta,
A. K. Iyer and U. Gupta,
Crit. Rev. Ther. Drug Carrier Syst.
,
2016,
33
, 159
–
193.
24 Y. Cui, X. Li, K. Zeljic, S. Shan, Z. Qiu and Z. Wang,
ACS Appl.
Mater. Interfaces
, 2019,
11
, 38190
–
38204.
25 C. Huang, C. Fu, Z.-P. Qi, W.-L. Guo, D. You, R. Li and Z. Zhu,
Artif. Cells, Nanomed., Biotechnol.
, 2020,
48
, 1010
–
1021.
26 H. Xue, Y. Ju, X. Ye, M. Dai, C. Tang and L. Liu,
Int. J. Biol.
Macromol.
, 2024,
254
, 128048.
27 Y. Luo, F. Xue, K. Liu, B. Li, C. Fu and J. Ding,
Mater. Des.
,
2021,
201
, 109484.
28 J. Wang, D. Li, C. Liang, C. Wang, X. Zhou, L. Ying, Y. Tao,
H. Xu, J. Shu, X. Huang, Z. Gong, K. Xia, F. Li, Q. Chen,
J. Tang and Y. Shen,
Small
, 2020,
16
(8), e1906415.
29 Y. Wang, M. Wu, L. Gu, X. Li, J. He, L. Zhou, A. Tong, J. Shi,
H. Zhu, J. Xu and G. Guo,
Drug Delivery
, 2017,
24
, 391
–
401.
30 F. Lin, Y. Liu, W. Luo, S. Liu, Y. Wang, R. Gu, W. Liu and
C. Xiao,
Int. J. Nanomed.
, 2022,
17
, 91
–
104.
31 J. Wang, D. Li, C. Liang, C. Wang, X. Zhou, L. Ying, Y. Tao,
H. Xu, J. Shu, X. Huang, Z. Gong, K. Xia, F. Li, Q. Chen,
J. Tang and Y. Shen,
Small
, 2020,
16
(8), e1906415.
32 L. Lei, Y. Bai, X. Qin, J. Liu, W. Huang and Q. Lv,
Gels
, 2022,
8
, 301.
33 M. Su, L. Ruan, X. Dong, S. Tian, W. Lang, M. Wu, Y. Chen,
Q. Lv and L. Lei,
Int. J. Biol. Macromol.
, 2023,
227
, 472
–
492.
34 Z. Jia, H. Zeng, X. Ye, M. Dai, C. Tang and L. Liu,
Heliyon
,
2023,
9
, e19933.
35 D. Silva, R. A. Sousa and A. J. Salgado,
Mater. Today Bio
, 2021,
9
, 100093.
36 B. Jeong, S. W. Kim and Y. H. Bae,
Adv. Drug Delivery Rev.
,
2012,
64
, 154
–
162.
37 A. Navaei, D. Truong, J. He
ff
ernan, J. Cutts, D. Brafman,
R. W. Sirianni, B. Vernon and M. Nikkhah,
Acta Biomater.
,
2016,
32
, 10
–
23.
38 X. Qin, Y. Xu, X. Zhou, T. Gong, Z.-R. Zhang and Y. Fu,
Acta
Pharm. Sin. B
, 2021,
11
, 835
–
847.
39 A. J. de Graaf, I. I. Azevedo Pr´
ospero dos Santos,
E. H. E. Pieters, D. T. S. Rijkers, C. F. van Nostrum,
T.
Vermonden,
R.
J.
Kok,
W.
E.
Hennink
and
E. Mastrobattista,
J. Controlled Release
, 2012,
162
, 582
–
590.
40 Y. Sun, Y. Bai, S. Liu, S. Cui and P. Xu,
IET Nanobiotechnol.
,
2023,
2023
, 1
–
9.
41 M. Xu, Y. Mou, M. Hu, W. Dong, X. Su, R. Wu and P. Zhang,
Asian J. Pharm. Sci.
, 2018,
13
, 373
–
382.
42 T. Yi, G. Zhuang and Y. Wang,
Arq. Bras. Med. Vet. Zootec.
,
2022,
74
, 641
–
648.
43 X. Chen, J. Chen, B. Li, X. Yang, R. Zeng, Y. Liu, T. Li,
R. J. Y. Ho and J. Shao,
J. Colloid Interface Sci.
, 2017,
490
,
542
–
552.
44 L. Li, Y. Li, C. Miao and R. Liu,
Adv. Polym. Technol.
, 2020,
2020
, 1
–
7.
45 X. Li, X. Xia, J. Zhang, M. Adu-Frimpong, X. Shen, W. Yin,
Q. He, W. Rong, F. Shi, X. Cao, H. Ji, E. Toreniyazov,
Q. Wang, J. Yu and X. Xu,
J. Pharm. Sci.
, 2023,
112
, 148
–
157.
46 D. Gao, S. Tang and Q. Tong,
Int. J. Nanomed.
, 2012,
7
, 3517
–
3526.
47 M. Adu
-
Frimpong, C. K. Firempong, E. Omari
-
Siaw,
Q. Wang, Y. M. Mukhtar, W. Deng, Q. Yu, X. Xu and J. Yu,
Drug Dev. Res.
, 2019,
80
, 230
–
245.
48 H. Zhang, Q. Wang, C. Sun, Y. Zhu, Q. Yang, Q. Wei, J. Chen,
W. Deng, M. Adu-Frimpong, J. Yu and X. Xu,
Pharmaceutics
,
2019,
11
, 107.
49 Y. Xue, Y. Liao, H. Wang, S. Li, Z. Gu, M. Adu
-
Frimpong,
J. Yu, X. Xu, H. D. C. Smyth and Y. Zhu,
J. Sci. Food Agric.
,
2023,
103
, 3628
–
3637.
50 L. Lu, Y. Wang, M. Cao, M. Chen, B. Lin, X. Duan, F. Zhang,
J. Mao, X. Shuai and J. Shen,
RSC Adv.
, 2017,
7
, 15041
–
15052.
51 Y. Mao, X. Li, G. Chen and S. Wang,
J. Pharm. Sci.
, 2016,
105
,
194
–
204.
52 M.-T. Sheu, H.-J. Jhan, C.-Y. Su, L.-C. Chen, C.-E. Chang,
D.-Z. Liu and H.-O. Ho,
Colloids Surf., B
, 2016,
143
, 260
–
270.
53 C. Al Sabbagh, J. Seguin, E. Agapova, D. Kramerich, V. Boudy
and N. Mignet,
Eur. J. Pharm. Biopharm.
, 2020,
157
, 154
–
164.
54 H. Li, X. Meng, Y. Zhang, M. Guo and L. Li,
Molecules
, 2023,
28
, 7319.
55 S. Kamiloglu, G. Sari, T. Ozdal and E. Capanoglu,
Food
Front.
, 2020,
1
, 332
–
349.
56 S. Li, J. Zhou, J. Zhang, D. Wang and J. Ma,
Saudi J. Biol. Sci.
,
2019,
26
, 2122
–
2126.
57 Y. D. Teng, H. Choi, R. C. Onario, S. Zhu, F. C. Desilets,
S. Lan, E. J. Woodard, E. Y. Snyder, M. E. Eichler and
R. M. Friedlander,
Proc. Natl. Acad. Sci. U. S. A.
, 2004,
101
,
3071
–
3076.
Nanoscale Adv.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Advances
Paper
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a

58 V. Krishna, H. Andrews, X. Jin, J. Yu, A. Varma, X. Wen and
M. Kindy,
J. Visualized Exp.
, 2013,
17
(78), 50111.
59 A. Singh, L. Krisa, K. L. Frederick, H. Sandrow-Feinberg,
S. Balasubramanian, S. K. Stackhouse, M. Murray and
J. S. Shumsky,
J. Neurosci. Methods
, 2014,
226
, 124
–
131.
60 J. Zhang, H. Ge, J. Li, L. Chen, J. Wang, B. Cheng and Z. Rao,
Regener. Ther.
, 2023,
24
, 180
–
189.
61 L. Luo, A. A. Albashari, X. Wang, L. Jin, Y. Zhang, L. Zheng,
J. Xia, H. Xu, Y. Zhao, J. Xiao, Y. He and Q. Ye,
Stem Cells Int.
,
2018,
2018
, 1
–
13.
62 H. Song, H. Geng, J. Ruan, K. Wang, C. Bao, J. Wang, X. Peng,
X. Zhang and D. Cui,
Nanoscale Res. Lett.
, 2011,
6
, 354.
63 Y.-C. Kuo and H.-W. Yu,
Colloids Surf., B
, 2011,
84
, 253
–
258.
64 T. N. S. Sulaiman, D. Larasati, A. K. Nugroho and S. Choiri,
Adv. Pharm. Bull.
, 2019,
9
, 382
–
392.
65 D.-L. Fang, Y. Chen, B. Xu, K. Ren, Z.-Y. He, L.-L. He, Y. Lei,
C.-M. Fan and X.-R. Song,
Int. J. Mol. Sci.
, 2014,
15
, 3373
–
3388.
66 Y. Lu, E. Zhang, J. Yang and Z. Cao,
Nano Res.
, 2018,
11
,
4985
–
4998.
67 O. A. A. Ahmed and S. M. Badr-Eldin,
Int. J. Pharm.
, 2020,
588
, 119778.
68 G. Soliman and F. Winnik,
Macromol. Biosci.
, 2010,
10
, 278
–
288.
69 A. D. Holmkvist, A. Friberg, U. J. Nilsson and J. Schouenborg,
Int. J. Pharm.
, 2016,
499
, 351
–
357.
70 L. Treuel, X. Jiang and G. U. Nienhaus,
J. R. Soc., Interface
,
2013,
10
, 20120939.
71 Q. Wang, N. Wei, X. Liu, A. Chang and K. Qian Luo,
Oncotarget
, 2017,
8
, 12013
–
12030.
72 L. Lei, Y. Bai, X. Qin, J. Liu, W. Huang and Q. Lv,
Gels
, 2022,
8
, 301.
73 V. Perumal, A. R. Ravula, A. Agas, A. Gosain, A. Aravind,
P. M. Sivakumar, S. S. I, K. Sambath, S. Vijayaraghavalu
and N. Chandra,
Brain Sci.
, 2023,
13
, 402.
74 X.-Q. Zheng, J.-F. Huang, J.-L. Lin, Y.-X. Zhu, M.-Q. Wang,
M.-L. Guo, X.-J. Zan and A.-M. Wu,
Colloids Surf., B
, 2021,
199
, 111532.
75 E. Dr´
aberov´
a, L. Del Valle, J. Gordon, V. Markov´
a,
B.
Smejkalov´
a,
L.
Bertrand,
J.
P.
de
Chadar´
evian,
D. P. Agamanolis, A. Legido, K. Khalili, P. Dr´
aber and
C. D. Katsetos,
J. Neuropathol. Exp. Neurol.
, 2008,
67
(4),
341
–
354.
76 V. Martinsen and P. Kursula,
Amino Acids
, 2022,
54
(1), 99
–
109.
77 D. Chung, A. Shum and G. Caraveo,
Front. Cell Dev. Biol.
,
2020,
8
, 567537.
78 Y. Hoshino, T. Okuno, D. Saigusa, K. Kano, S. Yamamoto,
H. Shindou, J. Aoki, K. Uchida, T. Yokomizo and N. Ito,
FASEB J.
, 2022,
36
(4), e22236.
79 A. Bernal and L. Arranz,
Cell. Mol. Life Sci.
, 2018,
75
(12),
2177
–
2195.
© 2024 The Author(s). Published by the Royal Society of Chemistry
Nanoscale Adv.
Paper
Nanoscale Advances
Open Access Article. Published on 16 September 2024. Downloaded on 9/27/2024 10:57:01 AM.
This article is licensed under a














